Become a Member to watch ad-free!
Relative expansion of solids, liquids and gases
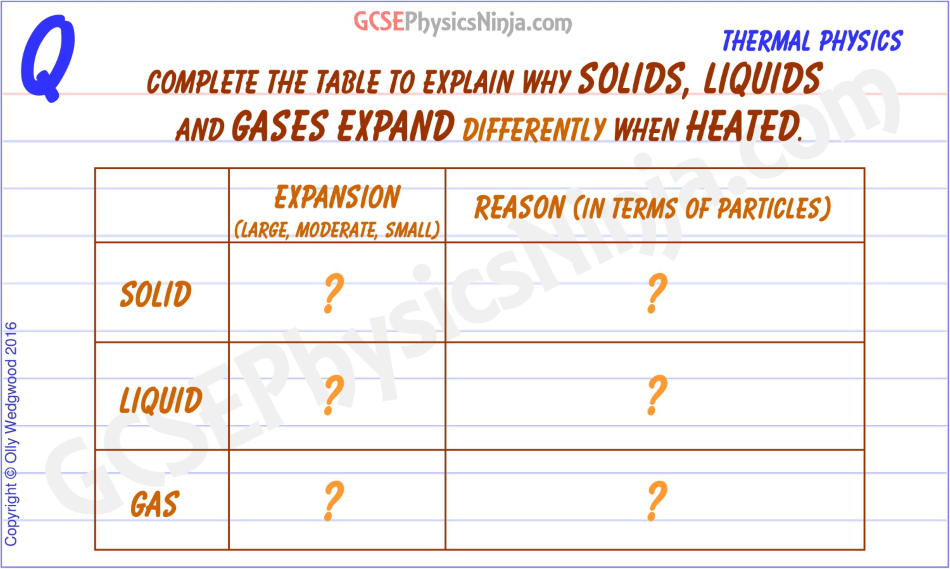
Solids, liquids and gases expand by different amounts when you heat them.
Solids only expand by a small amount. The reason for this is that the particles in a solid are held in place by very strong solid bonds. As you put thermal energy in by heating the solid, the kinetic energy of the particles increases, but the strong bonds don't let the particles move that far apart. Instead, the particles have larger vibrations about a fixed position.
In a liquid, the expansion is a little more than in solids.
The bonds in a liquid are weaker than in a solid, so as you heat up a liquid, the particles can move around each other faster and in so doing, move further apart.
Solids and liquids occupy a 'set' volume at a certain temperature. However, a gas will occupy the volume of whatever container it is in - i.e. a gas will fill the space available.
If you heat up a gas in a balloon, the added thermal energy means that the particles will fly around faster and so will collide with the walls of the balloon more frequently and with more force. This means that a hotter gas will have a higher pressure, which will make the balloon it is in expand (unless its walls are rigid as in a metal container, in which case it would be able to resist the increased gas pressure). The bonds between gas particles are very weak because they are so far apart.
All this means that gases expand much more than liquids or solids - if they are allowed!
Try this experiment - blow up a balloon at room temperature, then put it in the ice compartment of a fridge. What will happen to the balloon and why?
GCSE Keywords: bonds, spacing, particles, vibration
